Tryptamine Research Chemicals: A Complete Guide to 4-HO-MET, 4-AcO-DMT & More
Tryptamine Research Chemicals: A Complete Guide
Tryptamines represent one of the most structurally rich and pharmacologically interesting classes of psychoactive compounds studied in modern research chemistry. Sharing a core indole backbone with endogenous neurotransmitters like serotonin (5-HT) and DMT, synthetic tryptamine analogs offer researchers the opportunity to study serotonergic receptor systems at a level of granularity that scheduled compounds simply don't allow.
This guide covers the key tryptamine research chemicals currently available — their chemical properties, receptor binding profiles, duration, legal status across Europe, and harm reduction considerations.
What Are Tryptamine Research Chemicals?
Tryptamines are a class of compounds built around the tryptamine core — a bicyclic indole ring attached to an ethylamine chain. Modifications to the core structure (typically at the 4, 5, or N positions) produce compounds with markedly different pharmacological profiles.
The most well-researched endogenous tryptamine is DMT (N,N-dimethyltryptamine), which occurs naturally in humans and many plants. Synthetic analogs substitute or modify this structure to create compounds that are either legal alternatives for research or novel entities with distinct receptor profiles.
Key receptor targets for tryptamine research:
- 5-HT2A (primary target for psychedelic tryptamines)
- 5-HT2B and 5-HT2C (mood modulation, appetite)
- 5-HT1A (anxiolytic, autoreceptor function)
- Sigma-1 receptors (some N,N-dialkyl variants)
- Trace amine-associated receptors (TAARs)
Major Tryptamine Research Chemicals
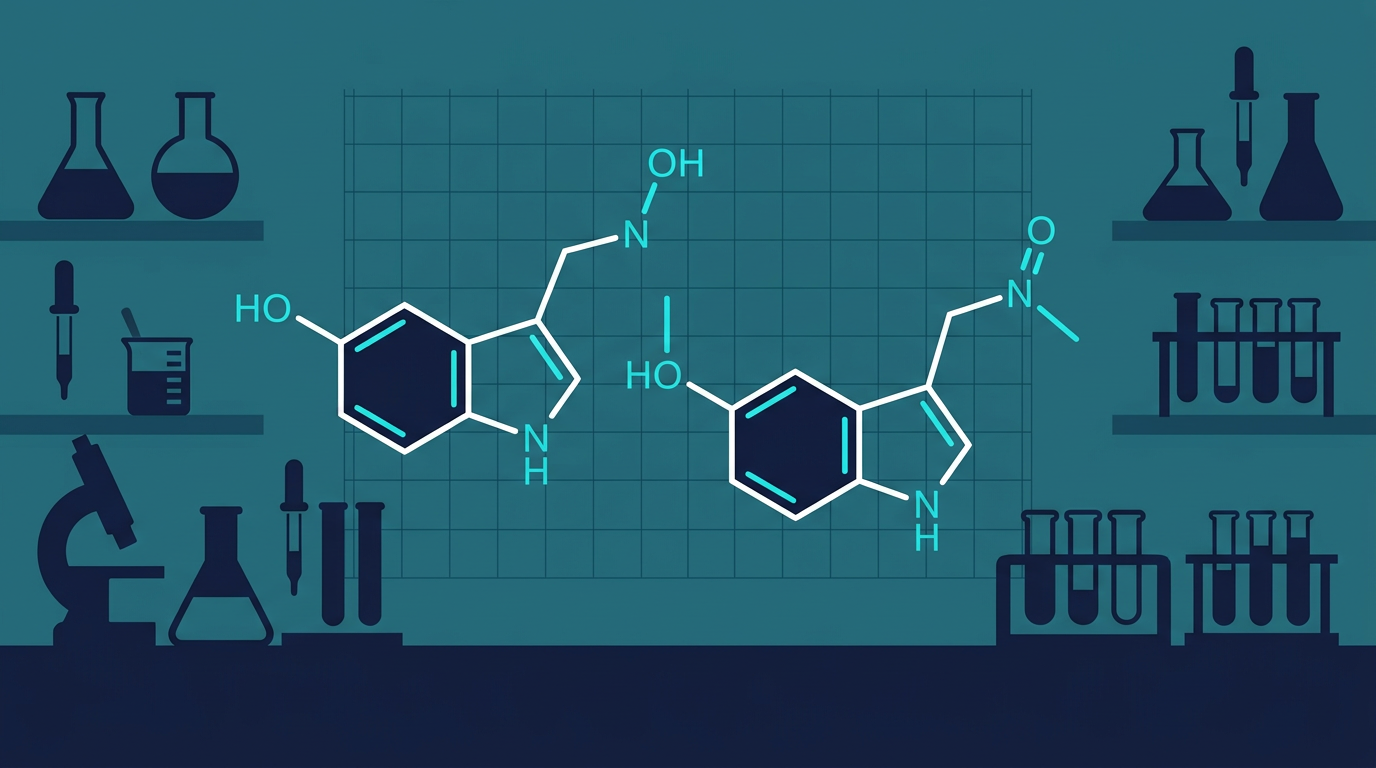
4-HO-MET (4-Hydroxy-N-methyl-N-ethyltryptamine)
CAS: 77872-43-6
Molecular Formula: C13H18N2O
Molecular Weight: 218.30 g/mol
4-HO-MET is a synthetic psychedelic tryptamine that acts primarily as a serotonin 5-HT2A receptor agonist. It is structurally similar to psilocin (4-HO-DMT) — the active metabolite of psilocybin — differing only in its N-substituents. This structural proximity makes it valuable for comparative receptor binding research.
Research Properties:
- Potency: Roughly equipotent to psilocin at 5-HT2A
- Duration: 4–6 hours (shorter than psilocybin's prodrug delay)
- Onset: 20–40 minutes
- Effect profile: Strong visual field activity, mood elevation, GABA modulation at higher doses
Available Forms: Fumarate salt (stable, crystalline), HCl salt
Research Applications: 5-HT2A agonism studies, comparative psilocin analogue research, receptor selectivity profiling
4-AcO-DMT (O-Acetylpsilocin)
CAS: 92292-84-7
Molecular Formula: C14H18N2O2
Molecular Weight: 246.31 g/mol
4-AcO-DMT (also known as synthetic mushroom compound or psilacetin) is the acetylated form of psilocin. It is widely considered a prodrug — upon administration it is deacetylated to psilocin by plasma and hepatic esterases. This makes it pharmacologically nearly identical to psilocybin's active metabolite, but with distinct pharmacokinetic properties.
Why Researchers Study 4-AcO-DMT:
- Allows study of psilocin pharmacodynamics without using Schedule I psilocybin
- Greater chemical stability than psilocin (does not oxidise as readily)
- Well-characterised deacetylation kinetics
- Useful for dose-controlled receptor occupancy studies
Research Properties:
- Potency: Equivalent to psilocin (bioavailability adjustment required)
- Duration: 4–6 hours
- Onset: 20–45 minutes
Available Forms: Fumarate salt (most stable), HCl salt
5-MeO-DALT (5-Methoxy-N,N-diallyltryptamine)
CAS: 928822-98-4
Molecular Formula: C15H20N2O
Molecular Weight: 244.33 g/mol
5-MeO-DALT occupies an interesting niche in tryptamine research — the 5-methoxy substitution and diallyl N-substitution produce a compound with a different receptor selectivity profile to most other tryptamines. It has meaningful affinity for 5-HT2A, 5-HT2B, and alpha-adrenergic receptors, making it relevant for research into multiple receptor systems simultaneously.
Research Properties:
- Duration: 2–4 hours (notably shorter than many tryptamines)
- Onset: Rapid — 10–20 minutes
- Receptor targets: 5-HT2A, 5-HT2B, alpha-1 adrenergic
Research Applications: Short-duration 5-HT2A agonism studies, alpha-adrenergic interaction research, receptor polypharmacology studies
4-HO-DiPT (4-Hydroxy-N,N-diisopropyltryptamine)
CAS: 14780-24-6
Molecular Formula: C16H24N2O
Molecular Weight: 264.38 g/mol
Known in the research literature as IPT or Isopropocybin, 4-HO-DiPT has a notably short duration profile (2–3 hours) despite structural similarity to longer-acting tryptamines. The bulky diisopropyl N-substitution is thought to affect receptor residence time. This makes it useful for research designs requiring defined, short exposure windows.
4-HO-EPT (4-Hydroxy-N-ethyl-N-propyltryptamine)
A more recently synthesised tryptamine analog, 4-HO-EPT is studied for its mixed N-substituent effects. Less characterised than the above compounds, making it of particular interest for novel pharmacology research.
Tryptamine Comparison Table
| Compound | 5-HT2A Activity | Duration | Onset | Key Research Use | |---|---|---|---|---| | 4-HO-MET | High (agonist) | 4–6 hrs | 20–40 min | Psilocin comparative studies | | 4-AcO-DMT | High (prodrug → psilocin) | 4–6 hrs | 20–45 min | Psilocybin mechanism research | | 5-MeO-DALT | Moderate (multi-receptor) | 2–4 hrs | 10–20 min | Multi-receptor profiling | | 4-HO-DiPT | High | 2–3 hrs | 20–30 min | Short-window agonism studies | | 4-HO-EPT | Under characterisation | 3–5 hrs | 20–40 min | Novel pharmacology |
Legal Status in Europe (March 2026)
Tryptamine legal status is highly jurisdiction-dependent. Most European countries use compound-specific scheduling rather than family-based laws, creating windows of legality for less-known analogs.
| Country | 4-HO-MET | 4-AcO-DMT | 5-MeO-DALT | Notes | |---|---|---|---|---| | Netherlands | Unscheduled | Unscheduled | Unscheduled | Research use permitted | | Germany | Controlled (NpSG) | Controlled (NpSG) | Check current list | Family-based scheduling applies | | Czech Republic | Unscheduled | Unscheduled | Unscheduled | Compound-specific list | | Poland | Check current list | Check current list | Check current list | Verify before ordering | | France | Broadly restricted | Broadly restricted | Broadly restricted | Analogue act applies | | Italy | Unscheduled (many) | Check | Unscheduled (likely) | Compound-specific | | UK | Class A | Class A | Class A | Psychoactive Substances Act 2016 |
Always verify current legal status in your jurisdiction before ordering. Laws change frequently and EuroChems only ships to countries where specific compounds are legal at time of order.
Storage and Handling
Tryptamine fumarate and HCl salts are generally stable under appropriate conditions:
- Temperature: Store at -20°C for long-term stability; 2–8°C for short-term (weeks)
- Light: Protect from UV exposure — indole compounds are photosensitive
- Humidity: Store in airtight containers with desiccant
- Container: Amber glass vials preferred for analytical reference samples
- Degradation indicator: Colour change (yellowing/browning) in psilocin-type compounds indicates oxidation
Safety and Research Protocols
All tryptamine research chemicals are labelled "For Research Purposes Only — Not for Human Consumption."
Analytical research considerations:
- Verify compound identity and purity via HPLC or GC-MS before use in assays
- Certificate of Analysis (COA) available for all EuroChems compounds
- Reagent testing (Ehrlich test produces purple for indole-containing tryptamines)
- Use appropriate PPE when handling powders (nitrile gloves, face mask)
- Weigh accurately — tryptamines are active at milligram scale
Conclusion
Tryptamines offer some of the most valuable receptor-system research tools available to EU researchers in 2026. Compounds like 4-HO-MET and 4-AcO-DMT provide access to psilocin/psilocybin pharmacology through legal channels in many European jurisdictions, while compounds like 5-MeO-DALT expand the research scope to multi-receptor systems.
EuroChems maintains a curated stock of tryptamine research chemicals with full COA documentation, discreet shipping, and compliance with destination-country laws.
Disclaimer: All information is provided for research and educational purposes only. EuroChems sells all products strictly for in vitro research and analytical purposes. Always verify the legal status in your jurisdiction before ordering.
Related Resources:
Related Topics
Explore Our Product Range
Browse our catalog of HPLC-verified research chemicals with guaranteed purity and fast EU shipping.
Shop Research Chemicals